Learn about 10 Difference between diamond and graphite
Welcome to a captivating journey into the world of carbon allotropes! Diamonds and graphite may both be made of carbon, but they are incredibly unique in their characteristics and uses. Join us as we uncover the distinct differences between these two intriguing forms of carbon. Diamonds and graphite may both be made of carbon, but they couldn’t be more different! Here are 10 key difference between these two fascinating forms of carbon diamond and graphite:
10 Difference between diamond and graphite
Diamonds and graphite, both made of carbon, have distinct 10 Difference between diamond and graphite:
Hardness: Diamonds are the hardest material (10 on the Mohs scale), while graphite is soft and can be easily marked.
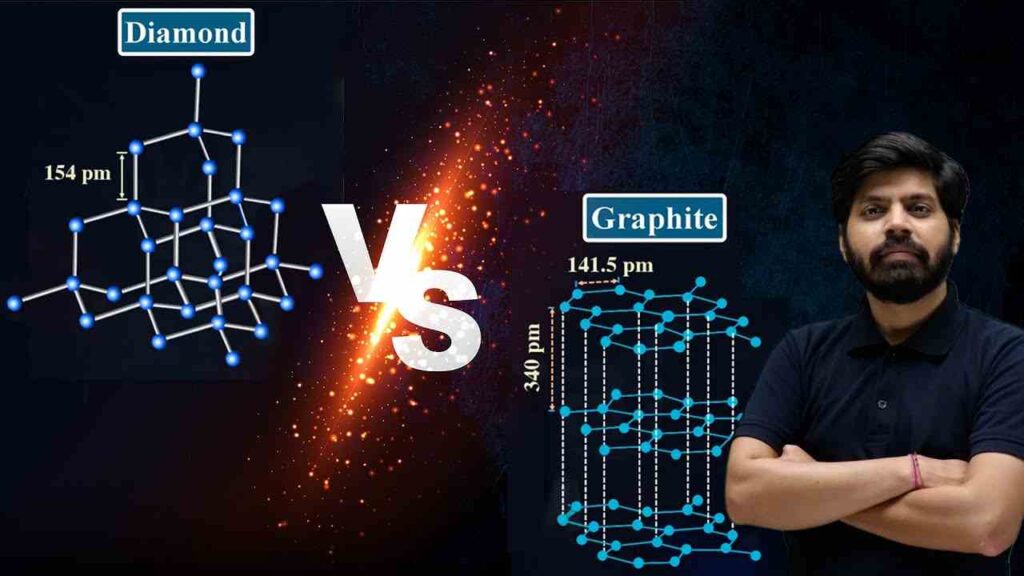
Structure: Diamonds have a three-dimensional lattice structure, while graphite has layered hexagonal rings.
Transparency: Diamonds are transparent, while graphite is opaque.
Also Read: Explore 10 Key Difference between Resistor and Resistance
Conductivity: Graphite is an excellent conductor of electricity, while diamonds are poor conductors.
Density: Diamonds are denser than graphite.
Melting Point: Diamonds have a higher melting point than graphite.
Uses: Diamonds are used as gemstones and in industrial applications, while graphite is used in pencils, lubricants, batteries, and nuclear reactors.
Cleavage: Diamonds have no cleavage, while graphite has perfect cleavage.
Also Read: Learn about 10 Difference between binary fission and multiple fission
Thermal Conductivity: Diamonds have extremely high thermal conductivity, while graphite has lower thermal conductivity.
Allotropes: Diamond and graphite are different forms of carbon with unique properties.
In summary, diamonds and graphite differ in their hardness, structure, transparency, conductivity, density, melting point, uses, cleavage, thermal conductivity, and allotropes. Despite being made of the same element, they have distinct characteristics!
Watch the video:
Also Read: Explore 10 Key Difference between potentiometer and voltmeter